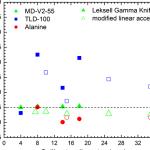
Connecting
Coordination compounds. like the FeCl4 – ion and CrCl3 6 NH3. are known as such simply because they contain ions or molecules linked, or coordinated, to some transition metal. They’re also referred to as complex ions or coordination complexes since they’re Lewis acidity-base complexes. The ions or molecules that bind to transition-metal ions to create these complexes are known as ligands (from Latin, to tie or bind). The amount of ligands certain to the transition metal ion is known as the coordination number.
Although coordination complexes are particularly significant within the chemistry from the transition metals, some primary group elements also form complexes. Aluminum, tin, and lead, for instance, form complexes like the AlF6 3-. SnCl4 2- and PbI4 2- ions.
Werner’s Theory of Coordination Complexes
Alfred Werner created a type of coordination complexs which is the next observations.
- A minimum of three different cobalt(III) complexes could be isolated when CoCl2 is dissolved in aqueous ammonia after which oxidized by air towards the +3 oxidation condition. A 4th complex can be created by slightly different techniques. These complexes have different colors and various empirical formulas.
- The reactivity from the ammonia during these complexes continues to be drastically reduced. Alone, ammonia reacts quickly with muriatic acidity to create ammonium chloride.
These complexes don’t interact with muriatic acidity, even at 100 o C.
- Solutions from the Cl – ion interact with Ag + ion to create a white-colored precipitate of AgCl.
When excess Ag + ion is put into solutions from the CoCl3 6 NH3 and CoCl3 5 NH3 H2 O complexes, three moles of AgCl are created for every mole of complex in solution, as may be expected.
However, only two Cl – ions within the CoCl3 5 NH3 complex and just among the Cl – ions in CoCl3 4 NH3 could be precipitated with Ag + ions.
- Measurements from the conductivity of aqueous solutions of those complexes claim that the CoCl3 6 NH3 and CoCl3 5 NH3 H2 O complexes dissociate in water to provide as many as four ions. CoCl3 5 NH3 dissociates to provide three ions, and CoCl3 4 NH3 dissociates to provide 3 ions.
Werner described these observations by suggesting that transition-metal ions like the Co 3+ ion possess a primary valence along with a secondary valence. The primary valence is the amount of negative ions required to fulfill the charge around the metal ion. In each one of the cobalt(III) complexes formerly described, three Cl – ions are necessary to fulfill the primary valence from the Co 3+ ion.
The secondary valence is the amount of ions of molecules which are coordinated towards the metal ion. Werner assumed the secondary valence from the transition metal during these cobalt(III) complexes is six. The formulas of those compounds can therefore be written the following.
Werner assumed that transition-metal complexes had definite shapes. Based on his theory, the ligands in six-coordinate cobalt(III) complexes are oriented toward the corners of the octahedron, as proven within the figure below.
Any ion or molecule with a set of nonbonding electrons could be a ligand. Many ligands are referred to as monodentate (literally, one-toothed) simply because they bite the metal in just one place.
Typical monodentate ligands receive within the figure below.
Other ligands can affix to the metal more often than once. Ethylenediamine (en) is really a typical bidentate ligand .
Each finish of the molecule contains a set of nonbonding electrons that may form a covalent bond to some metal ion. Ethylenediamine can also be one particualr chelating ligand. The word chelate develops from a Greek stem meaning claw. It’s accustomed to describe ligands that may carry the metal in several places, what sort of claw would.
Linking ethylene- diamine fragments gives tridentate ligands and tetradentate ligands. for example diethylenetriamine (dien) and triethylenetetramine (trien). Adding four -CH2 CO2 – groups for an ethylenediamine framework provides a hexadentate ligand. which could single-handedly fulfill the secondary valence of the transition-metal ion.
Transition-metal complexes happen to be characterised with coordination figures that vary from 1 to 12, but the most typical coordination figures are 2, 4, and 6. Types of complexes using these coordination figures receive within the table below.
Types of Common Coordination Figures
Lewis Acidity-Lewis base Method of Connecting in Complexes
G. N. Lewis was the first one to notice that the response from a transition-metal ion and ligands to create a coordination complex was similar towards the reaction between your H + and OH – ions to create water. The response between H + and OH – ions requires the donation of a set of electrons in the OH – ion towards the H + ion to create a covalent bond.
H + ion can be defined as an electron-pair acceptor. The OH – ion, however, is definitely an electron-pair donor. Lewis contended that any ion or molecule that behaves such as the H + ion ought to be an acidity. On the other hand, any ion or molecule that behaves such as the OH – ion ought to be basics. A Lewis acidity thus remains any ion or molecule that may accept a set of electrons. A Lewis base is definitely an ion or molecule that may donate a set of electrons.
When Co 3+ ions interact with ammonia, the Co 3+ ion accepts pairs of nonbonding electrons from six NH3 ligands to create covalent cobalt-nitrogen bonds as proven within the figure below.
The metal ion thus remains a Lewis acidity, and also the ligands coordinated for this metal ion are Lewis bases.
The Co 3+ ion is definitely an electron-pair acceptor, or Lewis acidity, since it has empty valence-covering orbitals you can use to carry pairs of electrons. To highlight these empty valence orbitals we are able to write the configuration from the Co 3+ ion the following.
There’s room within the valence covering of the ion for 12 more electrons. (Four electrons can be included to the threed subshell, two towards the 4s orbital, and 6 towards the 4p subshell.) The NH3 molecule is definitely an electron-pair donor, or Lewis base, because it features a set of nonbonding electrons around the nitrogen atom.
Based on this model, transition-metal ions form coordination complexes simply because they have empty valence-covering orbitals that may accept pairs of electrons from the Lewis base. Ligands must therefore be Lewis bases: They have to contain a minumum of one set of nonbonding electrons that may be donated to some metal ion.
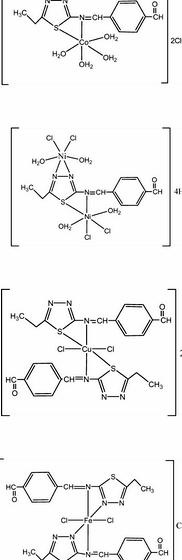
“One Schiff base ligand, bis(N,N-2-hyroxypropylenediaminesalicylaldimine), and it is metal complexes were synthesized and characterised within this research. The synthesis was achieved through the condensation of salicylaldehyde using the amine number of 1,3- diamino-2-propanol in methanol. The mononuclear and dinuclear complexes from Schiff base with tradition metals Cu(Il) and Ni(II) were made by refluxing in ethanol. Detailed structural portrayal from the ligand and it is complexes was performed through NMR, magnetic susceptibility, Ultra violet-Vis spectroscopy, IR spectroscopy, and Xray structure analysis.
At 70 degrees, the effective magnetic moment of mononuclear Cu(ll) complexes is 1.82BM confirming the existence of one unpaired electron. It’s distorted tetrahedral geometry and paramagnetic. The mononuclear Ni(Il) complex offers the effective magnetic moment, eff. 3.18BM which shows the existence of two unpaired electrons and paramagnetism. The magnetic qualities are described having a modified Bleaney-Bowers equation. This means a powerful antiferromagnetic exchange interaction between two copper atoms inside a dimeric complex.
It had been found with the X-ray structure analysis the mononuclear Cu(II) complex offers the triclinic lattice type and P-1 space group. The system cell parameters really are aEquals 7 .823(4)A b – = 9.688(5)A c = 10. 715( 6 )A = 91.56(1)° = 104.40(1)° and = 95.53(1)°. The very system of dinuclear Cu(II) complex is really a monoclinic lattice type and P2/c space group. The system cell parameters really are aEquals 18.084(2)A b – = 20.869(2)A c = 9.894(1)A and = 95.437(2)°”–Abstract, page iii.
Reddy, Prakash
Aronstam, Robert
M.S. in Chemistry
College of Missouri–Rolla
Includes bibliographical referencesIncludes bibliographical references (pages 73-78).
Thesis – Restricted Access
Crystals — Structure
Magnetic susceptibility
Metal complexes
Schiff bases
Electronic accessibility full-text of the document is fixed to Missouri S&T users. Otherwise, request this publication from Missouri S&T Library or contact check your local library.
Wang, Lili, “Synthesis and portrayal of some Schiff base metal complexes” (2006). Masters Theses. Paper 5906.
scholarsmine.mst.edu/masters_theses/5906




 Android locator map thesis writing
Android locator map thesis writing Into the world hsc thesis proposal
Into the world hsc thesis proposal Sample thesis proposal for inventory system
Sample thesis proposal for inventory system Thesis writing phd comics grading
Thesis writing phd comics grading Caution thesis writing in progress phd comics safety
Caution thesis writing in progress phd comics safety