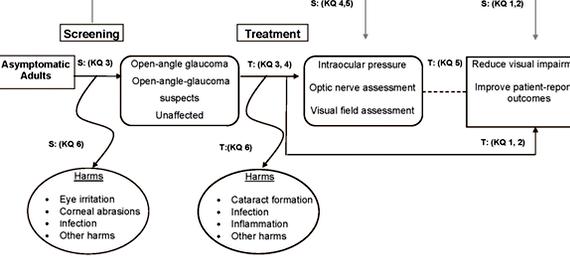
Glaucoma is a degenerative disease of the retina containing multifactorial structural and functional defects. There is a loss of retinal ganglion cells (RGCs) seen in this optic disorder. Cupping of the optic disk seen in glaucoma is a result of the loss of RGC axons at the optic nerve head where the nerve fibers connect. As a result of these factors, visual field loss is common among glaucoma patients. 1 In the United States, this visual field loss associated with glaucoma is one of the leading causes of blindness since glaucoma is difficult to detect. 2 Usually, increased intraocular pressure is associated with glaucoma as well as RGC loss. Lowering the IOP has not proved effective in reducing the visual field loss in all cases and the exact mechanism causing the RGC degeneration remains unknown. This makes it difficult to determine where treatments should be aimed.
However, there have been studies correlating an increase in excitotoxic amino acids, such as glutamate, in the vitreous humor with cell death occurring. 3 These studies support a proposal that excessive glutamate induces excitotoxicity which leads to apoptosis. This mechanism of glutamate-induced RGC death had been investigated to discover neuroprotective agents to prevent RGC loss. Studies of this nature have shown acetylcholine to be an effective neuroprotectant when applied previously before excitotoxic glutamate.
Acetylcholine binding to specifically α7 nAChr that are located on RGC has protective properties against RGC apoptosis. In order to study this, an α7 nAChR agonist has been used to activate the receptor. This agonist is PNU-282987 (developed by Pharmacia and Upjohn) and was used in previous studies to demonstrate its neuroprotectant effects in an in vivo rat model of glaucoma.
Previous studies have used fluorescently labeled antibodies against the glycoprotein Thy1.1 to observe the plasma membrane in RGCs. Previous results have indicated that the α7 nAChR agonist, PNU-282987 provides a neuroprotective effect against RGC loss in glaucoma induced conditions. However, others have demonstrated that there is down-regulation of the Thy 1.1 glycoprotein under certain conditions. As a result, another marker for RGCs is necessary. In this study, we used another fluorescent marker for an antibody against Brn3a, a nuclear marker that has also been shown to identify RGCs. In this study, both Thy1.1 and Brn3a markers were used to double label RGCs in glaucoma induced rats with and without application of PNU-282987. The neuroprotection effects of PNU-282987 on loss of RGCs were analyzed. The main objective of this study was to determine that application of PNU-282987 prevented the loss of RGCs associated with glaucoma and that the cells labeled using our techniques were indeed RGCs. Qualitative results were obtained, demonstrating that Brn3a labeled cells that were also labeled with Thy1.1. This indicated that there was no down-regulation of Thy1.1 prior to cell death and that RGCs could successfully be labeled with antibodies against Thy 1.1 and/or Brn3a. These qualitative results also strengthen past studies indicating that PNU-282987 has neuroprotective effects against the loss of RGCs.
Future quantitative results will be obtained to reinforce PNU-282987 as a neuroprotectant in the retina.
Rodriguez, Lucia, “Labeling of retinal ganglion cells in a rat glaucoma model” (2014). Honors Theses. Paper 2458.
scholarworks.wmich.edu/honors_theses/2458




 Sample thesis proposal business management
Sample thesis proposal business management Virginia woolf shakespeares sister thesis proposal
Virginia woolf shakespeares sister thesis proposal Declaration of authorship thesis proposal
Declaration of authorship thesis proposal Sample thesis proposal in english subject crossword
Sample thesis proposal in english subject crossword Architecture thesis title proposal for nursing
Architecture thesis title proposal for nursing