PDF
S.RAJA,__Durch,_090900002,_PhD_Thesis[2].pdf – Printed Version
Limited to Users only
Download (4MB) Request a duplicate
Abstract
The thesis handles the expansion and look for eco-friendly aqueous two phase systems made up of poly-ethylene-glycol (PEG) + sodium citrate (SC) + water to
 recover soluble proteins within the model tannery wastewater.
 Liquid- liquid equilibrium data of aqueous two-phase systems (ATPS) created by sodium citrate (SC) and poly-ethylene-glycol (PEG) of several molecular weights (600, 1000, 2000, 4000, and 6000) with water were developed at different pH (6, 7, and eight) and constant
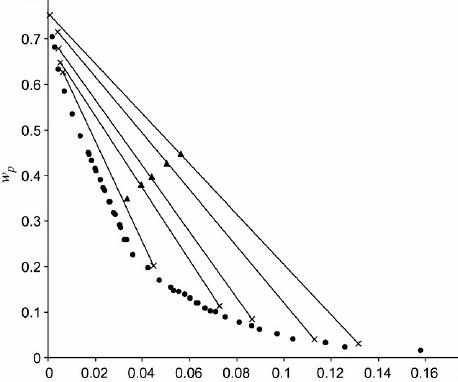
 temperature 25°C. Binodal data was described having a third order polynomial equation. A lift in molecular weight of PEG shifted the binodal curve to reduce PEG and salt
 concentrations. A lift in pH proven similar shape this will let you inclination to merge at high power PEG and salt. Tie-Line Length (TLL) along with the slope within the tie-line (STL)
 were connected while using equilibrium phase composition. The tie-lines were correlated when using the Othmer-Tobias and Bancroft equations along with the coefficient values were calculated. ATPS comprised of PEG 2000, 4000 and 6000 + SC was put on recoup the precious biomolecules within the model tannery wastewater system. Brought on by numerous protein
 partitioning parameters like PEG molecular weight (MW), power phase developing components, system pH, temperature and power sodium chloride were studied. Within the systems, the crude proteins preferentially partitioned for your salt wealthy bottom
 phase. Furthermore, the thermodynamic parameters as being a reason behind temperature were calculated for the PEG 6000/ SC system by using Van’t Hoff relationship.
It had been possible
 to extract 83.nearly for the finish phase upon an ATPS was comprised of PEG MW 6000 20% (w/w) / SC 15% (w/w) / .3M NaCl in the temperature of 30°C and pH 8. iii
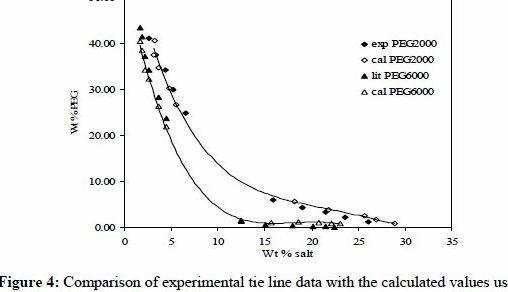
 ATPSs containing high molecular weight poly-ethylene glycol, PEG (10000) and biodegradable citrate salts (sodium citrate, potassium citrate and ammonium citrate) with water specified for to recoup the wastewater proteins within the model tannery wastewater system. The variations within the phase diagram were described based on ionic radius of cations, Gibbs free energy of hydration (Ghydation) of cations and efficient excluded volume (EEV) of salts. The salting out capacity within the cations adopted the succession: Sodium citrate Potassium citrate Ammonium citrate. Setschenow type equations were selected to correlate tie-line compositions. During partitioning studies, recovery of tannery wastewater proteins in PEG 10000 + Sodium citrate ATPS was discovered a lot better than others. It had been easy to recover 95.86% of proteins from tannery wastewater for the finish phase with PEG 10000 30% (w/w) + Sodium citrate 13% (w/w) at 30C. The partition coefficients were correlated while using the salt compositions getting a quadratic equation along with the coefficients were calculated. Enhanced recovery of proteins was attempted with PEG 6000 +SC +Water system using response surface methodology (RSM).
During this method, a consecutive optimization strategy (MINITAB – 15.) which incorporated fractional factorial design (fFD) and central composite design (CCD) were selected. Five factors namely, power PEG 6000, power SC, pH within the system, power NaCl and temperature were selected because the factors affecting the protein partitioning. The fFD states only pH, power NaCl and temperature were the important factors. These 4 elements were further enhanced by using CCD experiments. By using this strategy, another order polynomial model was acquired for the protein recovery and it also was validated. The optimum recovery was discovered as 93.46% when pH, NaCl concentration and temperature were stored at 7.5, .1 M and 33C
 iv correspondingly, for almost any phase system made up of 20% (w/w) PEG 6000 + 15% (w/w) SC. Thus the suggested ATPS can serve rather in the traditional precipitation
 approach to recover the soluble proteins from tannery wastewater.
Thesis (Phd. Thesis)




 Preliminary pages of thesis writing
Preliminary pages of thesis writing The glass menagerie essay thesis proposal
The glass menagerie essay thesis proposal Sachin katti phd thesis writing
Sachin katti phd thesis writing Research methods and thesis writing 2007 ed. pdf
Research methods and thesis writing 2007 ed. pdf Cooperative mimo phd thesis writing
Cooperative mimo phd thesis writing