* Final gross prices can vary based on local VAT.
Abstract
Solid fat nanoparticles (SLN) came to be in 1991 like a delivery system mixing benefits of other nanoparticulate systems and simultaneously staying away from/minimizing a few of their limitations. In 1999/2000 the 2nd generation of nano(structured) fat carriers (NLC) was created, showing improved qualities for example elevated drug loading and physical stability. Variations towards the first generation are discussed. Large-scale production, incorporation of NLC concentrates in final dermal products, and recognized regulatory status of excipients – as prerequisites for market introduction – are presented. Special emphasis can also be provided to the issue of nanotoxicity/nanotolerability, thinking about the growing concern within the public perception. The mechanisms of action are presented. As the most crucial qualifying criterion for that performance of the delivery system, in vivo data within the literature are reviewed. An introduction to marketed dermal products (cosmetics) is offered and also the future for dermal pharma products and cosmetics forecasted.
Keywords
Fat nanoparticles Nanostructured fat carriers NLC SLN Transmission enhancement Dermal products Large-scale production Nanotoxicity Regulatory status In vivo performance Ruthless homogenization
References
Almeida AJ, Runge S, Mller RH (1997) Peptide-loaded solid fat nanoparticles (SLN): influence of production parameters. Int J Pharm 149(2):255–265 CrossRef
Baiseng N (2013) Ultra-small Nanostructured Fat Carriers (usNLC). PhD-thesis, Freie Universitt Berlin (when preparing)
Bargoni A, Cavalli R, Zara GP, Fundaro A, Caputo O, Gasco MR (2001) Transmucosal transport of tobramycin incorporated in solid fat nanoparticles (SLN) after duodenal administration to rats.
Part II–tissue distribution. Pharmacol Res 43(5):497–502 PubMed CrossRef
Betzig E, Patterson GH, Sougrat R, Lindwasser OW, Olenych S, Bonifacino JS et al (2006) Imaging intracellular fluorescent proteins at nanometer resolution. Science 313(5793):1642–1645 PubMed CrossRef
Bhaskar K, Anbu J, Ravichandiran V, Venkateswarlu V, Rao Y (2009a) Fat nanoparticles for transdermal delivery of flurbiprofen: formulation, in vitro. ex vivo as well as in vivo studies. Lipids Health Dis 8:6 PubMed PubMedCentral CrossRef
Bhaskar K, Mohan CK, Lingam M, Mohan SJ, Venkateswarlu V, Rao YM et al (2009b) Growth and development of SLN and NLC enriched hydrogels for transdermal delivery of nitrendipine: in vitro as well as in vivo characteristics. Drug Dev Ind Pharm 35(1):98–113 PubMed CrossRef
Bunjes H, Westsen K, Koch MHJ (1996) Crystallization inclination and polymorphic transitions in triglyceride nanoparticles. Int J Pharm 129(1–2):159–173 CrossRef
Cavalli R, Gasco MR, Chetoni P, Burgalassi S, Saettone MF (2002) Solid fat nanoparticles (SLN) as ocular delivery system for tobramycin. Int J Pharm 238(1–2):241–245 PubMed CrossRef
del Pozo-Rodriguez A, Pujals S, Delgado D, Solinis MA, Gascon AR, Giralt E et al (2009a) A proline-wealthy peptide improves cell transfection of solid fat nanoparticle-based non-viral vectors. J Control Release 133(1):52–59 PubMed CrossRef
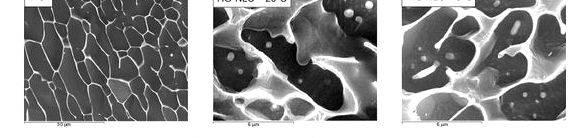
del Pozo-Rodriguez A, Solinis MA, Gascon AR, Pedraz JL (2009b) Short- and lengthy-term stability study of lyophilized solid fat nanoparticles for gene therapy. Eur J Pharm Biopharm 71(2):181–189 PubMed CrossRef
Domb AJ (1993) Lipospheres for controlled delivery of drugs. US Patent 5,188,837
EU (2011) European Commission Recommendation on the phrase nanomaterial, Regulation (Ec) No. 1223/2009 from the european parliament as well as the council on cosmetics, L 342/59 (2009).
Gasco MR (1993) Way of producing solid fat microspheres getting a narrow size distribution. US Patent 5,250,236
Gasco MR (2002) Microparticles for drug delivery across mucosa and also the blood–brain barrier. US Patent 6,419,949
Gasco MR (2007) Fat nanoparticles: perspectives and challenges. Adv Drug Deliv Rev 59(6):377–378 PubMed CrossRef
Hess HF (2007) Far-field optical nanoscopy. Science 316(5828):1153–1158 CrossRef
Hess SM, Girirajan TPK, Mason MD (2006) Ultra-high res imaging by fluorescence photoactivation localization microscopy. Biophys J 91(11):4258–4272 PubMed PubMedCentral CrossRef
Jenning V (1999) Feste Fat-Nanopartikel (SLN®) als Trgersystem fr die dermale Applikation von Retinol: Wirkstoffinkorporation, freisetzung und Struktur (Solid fat nanoparticles (SLN®) as carrier for that dermal use of Retinol: drug incorporation, release and structure from the carrier). PhD-thesis, Freie Universitt Berlin
Jenning V, Gohla S (2001) Encapsulation of retinoids in solid fat nanoparticles (SLN). J Microencapsul 18(2):149–158 PubMed CrossRef
Jores K, Mehnert W, Drechsler M, Bunjes H, Johann C, Mder K (2004) Investigations around the structure of solid fat nanoparticles (SLN) and oil-loaded solid fat nanoparticles by photon correlation spectroscopy, field-flow fractionation and transmission electron microscopy. J Control Release 95(2):217–227 PubMed CrossRef
Joshi M, Patravale V (2006) Formulation and look at nanostructured fat carrier (NLC)-based gel of valdecoxib. Drug Dev Ind Pharm 32(8):911–918 PubMed CrossRef
Keck CM, Baisaeng N, Durand P, Prost M, Meinke MC, Muller RH (2014) Oil-enriched, ultra-small nanostructured fat carriers (usNLC): A manuscript delivery system according to switch-flop structure. Int J Pharm 477(1–2):227–235
Keck CM, Mller RH (2010) Silber-Fat-Zwerge – Ein neues Therapiekonzept gegen Neurodermitis. Quantos 1:6–11
Keck CM, Mller RH (2013) Nanotoxicological classification system (NCS) – helpful information for that risk-benefit assessment of nanoparticulate drug delivery systems. Eur J Pharm Biopharm 84(3):445–448 PubMed CrossRef
Keck CM, Schwabe K (2009) Silver-nanolipid complex for application to atopic eczema skin: rheological portrayal, in vivo efficiency and theory of action. J Biomed Nanotechnol 5(4):428–436 PubMed CrossRef
Keck CM, Schwabe K, Rimpler C (2008) Fat nanoparticles for that enhancement from the dermal action of solid particles. European Patent Application No 08 019 3063, 4 November 2008
Keck CM, Boreham T, Schlieter D, Peters D, Mller RH, Alexiev U (2013) Nanostructures and drug distribution within Nanostructured Fat Carriers (NLC). Annual meeting of controlled release society, Honululu, p ID 100536
Kim T-Y, Uji-i H, Moeller M, Muls B, Hofkens J, Alexiev U (2009) Monitoring the interaction of merely one G-protein key binding site with rhodopsin disk membranes upon light activation. Biochemistry 48(18):3801–3803 PubMed CrossRef
Kim T-Y, Schlieter T, Haase S, Alexiev U (2012) Activation and molecular recognition from the GPCR rhodopsin – insights from time-resolved fluorescence depolarisation and single molecule experiments. Eur J Cell Biol 91(4):300–310 PubMed CrossRef
Kirchberg K, Kim T-Y, Haase S, Alexiev U (2010) Functional inter-action structures from the photochromic retinal protein rhodopsin. Photochem Photobiol Sci 9(2):226–233 PubMed CrossRef
Kchler S, Radowski MR, Blaschke T, Dathe M, Plendl J, Haag R et al (2009) Nanoparticles for skin transmission enhancement–a comparison of the dendritic core-multishell-nanotransporter and solid fat nanoparticles. Eur J Pharm Biopharm 71(2):243–250 PubMed CrossRef
Lucks JS, Mller RH (1996) Inventors Excipient of solid fat particles (solid fat nanospheres (SLN)), Air 0605497 B1
Maia CS, Mehnert W, Schfer-Korting M (2000) Solid fat nanoparticles as drug carriers for topical glucocorticoids. Int J Pharm 196(2):165–167 PubMed CrossRef
Maia CS, Mehnert W, Schaller M, Korting HC, Gysler A, Haberland A et al (2002) Drug targeting by solid fat nanoparticles for dermal use. J Drug Target 10(6):489–495 CrossRef
Masters DB, Domb AJ (1998) Liposphere local anesthetic timed-release for perineural site application. Pharm Res 15(7):1038–1045 PubMed CrossRef
Miglietta A, Cavalli R, Bocca C, Gabriel L, Gasco MR (2000) Cellular uptake and cytotoxicity of solid fat nanospheres (SLN) incorporating doxorubicin or paclitaxel. Int J Pharm 210(1–2):61–67 PubMed CrossRef
Mitri K, Shegokar R, Gohla S, Anselmi C, Mller RH (2011) Fat nanocarriers for dermal delivery of lutein: preparation, portrayal, stability and gratifaction. Int J Pharm 414(1–2):267–275 PubMed CrossRef
Mller BW (1998) Mikroemulsionen als neue Wirkstofftrgersysteme. In: Mller RH, Hildebrand GE (eds) Pharmazeutische Technologie: Moderne Arzneiformen. Wissenschaftliche Verlagsgesellschaft, Stuttgart, pp 161–169
Mller R, Mehnert W, Lucks JS, Schwarz C, Zur Mhlen A, Meyhers H et al (1995) Solid fat nanoparticles (SLN): an alternate colloidal carrier system for controlled drug delivery. Eur J Pharm Biopharm 41(1):62–69
Mller RH, Jenning V, Mder K, Lippacher A (2000a) Fat particles based on mixtures of liquid and solid lipids and also the way of producing same. PCT/EP2000/004112
Mller RH, Jenning V, Mder K, Lippacher A (2000b) Lipidpartikel auf der Basis von Mischungen von flssigen und festen Lipiden und Verfahren zu ihrer Herstellung. EP1176949A2
Mller RH, Mder K, Gohla S (2000c) Solid fat nanoparticles (SLN) for controlled drug delivery – overview of the condition from the art. Eur J Pharm Biopharm 50(1):161–177 PubMed CrossRef
Mller RH, Radtke M, Wissing SA (2002a) Nanostructured fat matrices for improved microencapsulation of medication. Int J Pharm 242(1–2):121–128 PubMed CrossRef
Mller RH, Radtke M, Wissing SA (2002b) Solid fat nanoparticles (SLN) and nanostructured fat carriers (NLC) in cosmetic and dermatological formulations. Adv Drug Deliv Rev 54(Suppl 1):S131–S155 PubMed CrossRef
Mller RH, Petersen RD, Hommoss A, Pardeike J (2007) Nanostructured fat carriers (NLC) in cosmetic dermal products. Adv Drug Deliv Rev 59(6):522–530 PubMed CrossRef
Mller RH, Gohla S, Keck CM (2011a) Condition of the skill of nanocrystals – special features, production, nanotoxicology aspects and intracellular delivery. Eur J Pharm Biopharm 78(1):1–9 PubMed CrossRef
Mller RH, Shegokar R, Keck CM (2011b) two decades of fat nanoparticles (SLN and NLC): present condition of development and industrial applications. Curr Drug Discov Technol 8(3):207–227 PubMed CrossRef
Pardeike J, Mller RH (2007) Coenzyme Q10 Supplement-loaded NLCs: preparation, occlusive qualities and transmission enhancement. Pharm Tech Eur 19(7):46–49
Pardeike J, Hommoss A, Mller RH (2009) Fat nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int J Pharm 366(1–2):170–184 PubMed CrossRef
Pardeike J, Schwabe K, Mller RH (2010) Influence of nanostructured fat carriers (NLC) around the physical qualities from the Cutanova Nanorepair Q10 cream and also the in vivo skin hydration effect. Int J Pharm 396(1–2):166–173 PubMed CrossRef
Pople PV, Singh KK (2006) Development and look at topical formulation that contains solid fat nanoparticles of vit a. AAPS PharmSciTech 7(4):91 PubMed CrossRef
Puglia C, Blasi P, Rizza L, Schoubben A, Bonina F, Rossi C et al (2008) Fat nanoparticles for prolonged topical delivery: an in vitro as well as in vivo analysis. Int J Pharm 357(1–2):295–304 PubMed CrossRef
Radtke M (2003) Grundlegende Untersuchungen zur Arzneistoffinkorporation, freisetzung und Struktur von SLN und NLC. Ph.D. thesis, Freie Universitt Berlin, Berlin
Rogiers V (2001) EEMCO guidance for that assessment of transepidermal water reduction in cosmetic sciences. Skin Pharmacol Appl Skin Physiol 14:117–128
Rust MJ, Bates M, Zhuang X (2006) Sub-diffraction-limit imaging by stochastic optical renovation microscopy (STORM). Nat Methods 3(10):793–795 PubMed PubMedCentral CrossRef
Schfer-Korting M, Mehnert W, Korting H-C (2007) Fat nanoparticles for improved topical use of drugs for skin illnesses. Adv Drug Deliv Rev 59(6):427–443 PubMed CrossRef
Schubert MA, Mller-Goymann CC (2005) Characterisation of surface-modified solid fat nanoparticles (SLN): influence of lecithin and nonionic emulsifier. Eur J Pharm Biopharm 61(1–2):77–86 PubMed CrossRef
Schwarz JC, Weixelbaum A, Pagitsch E, Low M, Resch GP, Valenta C (2012) Nanocarriers for dermal drug delivery: influence of preparation method, carrier type and rheological qualities. Int J Pharm 437(1–2):83–88 PubMed CrossRef
Sinambela P, Shegokar R, Gohla S, Mller RH (2011) Dermal nanostructured fat carriers (NLC) – the invisible patch. AAPS annual meeting, Washington Electricity, p T2160
Souto EB, Wissing SA, Barbosa CM, Mller RH (2004) Growth and development of a controlled release formulation according to SLN and NLC for topical clotrimazole delivery. Int J Pharm 278(1):71–77 PubMed CrossRef
Teeranachaideekul V, Boonme P, Souto EB, Mller RH, Junyaprasert VB (2008) Influence of oil content on physicochemical qualities and skin distribution of Earth red-loaded NLC. J Control Release 128(2):134–141 PubMed CrossRef
ner M, Wissing SA, Yener G, Mller RH (2005a) Skin moisturizing effect and skin transmission of ascorbyl palmitate entrapped in solid fat nanoparticles (SLN) and nanostructured fat carriers (NLC) integrated into hydrogel. Pharmazie 60(10):751–755 PubMed
ner M, Wissing SA, Yener G, Mller RH (2005b) Solid fat nanoparticles (SLN) and nanostructured fat carriers (NLC) for use of ascorbyl palmitate. Pharmazie 60:577–582 PubMed
US Fda (Food and drug administration) (2011) Draft guidance, thinking about whether an Food and drug administration-controlled product involves the use of nanotechnology, Guidance for Industry
von Ngeli CW (1893) ber oligodynamische Erscheinungen in lebenden Zellen. Neue Denksch 33:1–51
Zara GP, Bargoni A, Cavalli R, Fundaro A, Vighetto D, Gasco MR (2002) Pharmacokinetics and tissue distribution of idarubicin-loaded solid fat nanoparticles after duodenal administration to rats. J Pharm Sci 91(5):1324–1333 PubMed CrossRef
zur Mhlen A, Schwarz C, Mehnert W (1998) Solid fat nanoparticles (SLN) for controlled drug delivery–drug release and release mechanism. Eur J Pharm Biopharm 45(2):149–155 PubMed CrossRef




 Operational risk management in banks thesis proposal
Operational risk management in banks thesis proposal Reconfigurable manufacturing system thesis proposal
Reconfigurable manufacturing system thesis proposal Uppsala university phd thesis proposal
Uppsala university phd thesis proposal Entrepreneurs born or made dissertation proposal
Entrepreneurs born or made dissertation proposal Master thesis proposal finance department
Master thesis proposal finance department